Copyright © 2025 Novo Surgical Inc. All Rights Reserved
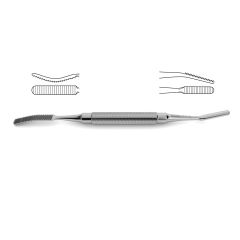
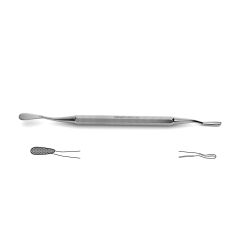
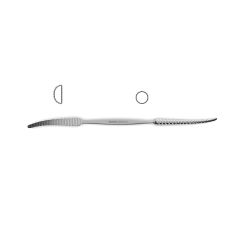
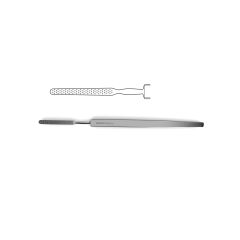
Bone File #33
double-ended, plain serrations, 6.0 mm & 5.0 mm wide ends, 7" (17.5 cm)
Details
Novo Surgical’s Bone File #33 is ideally suit for use in neurosurgical and orthopedic surgeries that require manipulation of bone in and around delicate tissue structures. This type of instrument can be useful in filing down the skull in procedures like craniotomies or in reshaping the vertebral arch during laminectomies or hemi-laminectomies. This gives added protection to the brain tissue or neurovascular bundles that can pass over these cut surfaces that may be jagged and could cause additionally injury or trauma. Additionally this file is double ended with plain serrations and features a 6.0 mm and 5.0 mm wide ends.
Please note that website images are often representative of the product family and may not conform to the exact specifications or features of the item you select. The final specifications will match the description of the specific catalog item you select. Please review such specifications in the item description to ensure the correct product is ordered.
More Information
| Manufacturer | Novo Surgical Inc. |
|---|---|
| Grade | Premium Operating Room |
| Material Finish | Stainless Steel |
| Obsolete Item# | 7720-11 |
| Specialty | NEURO, ORTHO & SPINE/RASPS & FILES |
| Sterility | Non-Sterile |
| Unit Of Measurement | Each |
| Lead Time | 8-15 Weeks |
| Usage | Reusable |
| Countries for Sale | USA, CANADA |
Warranty
Novo Surgical instruments are guaranteed to be free from defects in materials and workmanship when used under normal conditions for their intended surgical purpose. Any Novo Surgical instrument that is determined to be defective will, at Novo Surgical's discretion, be repaired or replaced. Normal wear and tear and/or instrument misuse, including improper use or inadequate maintenance, are not covered under the manufacturer's warranty. All Novo Surgical instruments are warranted only to the original purchaser. This instrument is intended to be used only for the specific purpose for which it was designed. It is the responsibility of clinicians and sterilization professionals to familiarize themselves with proper techniques for using and caring for this instrument. For more detailed instrument care recommendations, please visit the Instrument Care section of our website.
The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. By purchasing this product, you warrant that you are either a medical practitioner or purchasing on behalf of a licensed medical facility, educational or government agency, or a customer that sells these products to such institutions. After your order is placed, we may request you to provide additional information, including, but not limited to, the name and address of healthcare facility or the National Provider Identifier (NPI) number for the provider you are ordering on behalf of. Please note that the lack of a satisfactory response may result in the delay or cancellation of your order.